Quantum BioPharma Submits Investigational New Drug (IND) Application to United States Food and Drug Administration (FDA) for Lucid-MS Phase Two Clinical Trial in Multiple Sclerosis
TORONTO, April 01, 2026 (GLOBE NEWSWIRE) -- Quantum BioPharma Ltd. (NASDAQ: QNTM) (CSE: QNTM) (FRA: 0K91) (“Quantum BioPharma” or the “Company”), a biopharmaceutical company dedicated to building a portfolio of innovative assets and biotech solutions, today announced that it has formally submitted an Investigational New Drug (IND) application to the U.S. Food and Drug Administration (FDA) for Lucid-21-302 (Lucid-MS), its new drug candidate for the treatment of multiple sclerosis (MS). This milestone IND submission supports Quantum’s planned Phase 2 clinical trial evaluating its first-in-class therapeutic treatment targeting demyelination, advancing Quantum BioPharma’s strategic growth in the global neurological market.
Milestone IND submission supports planned phase-2 clinical trial evaluating First-in-Class therapeutic treatment targeting demyelination to advance Quantum BioPharma’s strategic growth in global neurological market.
This strategic milestone represents a significant advancement in Quantum BioPharma's clinical development pipeline and underscores the Company's commitment to delivering innovative therapeutic solutions for patients suffering from debilitating neurological conditions.
Comprehensive IND Submission and Development Timeline
The IND submission includes data from nonclinical pharmacology and toxicology studies, along with manufacturing and quality information, to support initiation of the planned Phase 2 clinical trial. Subject to FDA clearance, Quantum BioPharma intends to initiate the Phase 2 study as soon as possible.
| Phase 1 Clinical Trials | Completed | ✓ Safe & Well-Tolerated |
| IND Submission to FDA | March 2026 | ✓ Submitted |
| Anticipated FDA Response | Q2 2026 | Pending |
| Phase 2 Trial Initiation | Q2 2026 | Planned |
| Phase 2 Interim Data | Q4 2026 | Projected |
First-in-Class Therapeutic Innovation
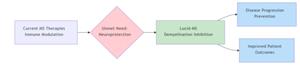
Lucid-MS is designed to provide neuroprotection through the inhibition of demyelination—a key driver of disease progression in MS. This innovative mechanism represents a differentiated therapeutic approach in the global MS market, where many existing therapies primarily focus on modulating the immune system rather than addressing the underlying neurodegeneration.
The planned Phase 2 trial will evaluate the efficacy, safety, and tolerability of Lucid-MS in people with MS. In prior Phase 1 clinical trials, Lucid-MS demonstrated a favorable safety profile and was well-tolerated in healthy participants providing a strong foundation for continued development.
“This IND submission is supported by a comprehensive preclinical research package designed to characterize the safety profile of Lucid-MS and support its evaluation in patients. The data we've compiled reflects many years of dedicated scientific research. We look forward to working with the FDA as we progress toward clinical evaluation of this novel approach to prevent disease progression in MS," said Dr. Andrzej Chruscinski, Vice-President, Scientific and Clinical Affairs at Quantum BioPharma.
Strategic Market Opportunity
Multiple sclerosis affects approximately 2.8 million people worldwide (source: https://pmc.ncbi.nlm.nih.gov Atlas of MS, Third Edition (PMC/NCBI)), representing a significant global healthcare challenge and substantial market opportunity for innovative treatments. The MS therapeutic market is projected to exceed $38 billion by 2030 according to Grand View Research www.grandviewresearch.com, driven by increasing diagnosis rates and demand for disease-modifying therapies.
Quantum BioPharma's differentiated approach—targeting demyelination directly—positions Lucid-MS to address unmet patient needs and potentially capture meaningful market share in this growing segment.
Zeeshan Saeed, CEO of Quantum BioPharma added, “The submission of our IND marks an important milestone in the advancement of Lucid-MS into clinical development for multiple sclerosis. We believe our approach targeting demyelination represents a differentiated strategic position in a field where significant innovation is needed. This milestone reflects our commitment to shareholder value creation through disciplined investment in high-potential therapeutic programs.”
“Our approach of combining clinical development with consumer health products like unbuzzd, targets sustainable revenue streams that support continued research and development operational cash without shareholder dilution. We remain focused on executing our growth strategy while advancing potentially transformative treatments for MS patients in need."
Quantum BioPharma’s Diversified Business Platform
Quantum BioPharma's diversified business platform targets growth through multiple potential value drivers:
| Clinical Development | Lucid-MS Phase 2 trial | High-value therapeutic innovation |
| Consumer Health | UNBUZZD™ rapid alcohol metabolizer | Revenue generation via 7% royalty |
| Strategic Investments | FSD Strategic Investments Inc. | Digital assets |
About Quantum BioPharma Ltd.
Quantum BioPharma is a biopharmaceutical company dedicated to building a portfolio of innovative assets and biotech solutions for the treatment of challenging neurodegenerative and metabolic disorders and alcohol misuse disorders with drug candidates in different stages of development. Through its wholly owned subsidiary, Lucid Psycheceuticals Inc. (“Lucid”), Quantum BioPharma is focused on the research and development of its lead compound, Lucid-MS. Lucid-MS is a patented new chemical entity shown to prevent and reverse myelin degradation, the underlying mechanism of multiple sclerosis, in preclinical models. Quantum BioPharma invented UNBUZZD™ and spun out its OTC version to a company, Unbuzzd Wellness Inc. (“UWI”), led by industry veterans. Quantum BioPharma retains ownership of 19.84% (as of December 31, 2025) of UWI at www.unbuzzd.com. The agreement with UWI also includes royalty payments of 7% of sales from unbuzzd™ until payments to Quantum BioPharma total $250 million. Once $250 million is reached, the royalty drops to 3% in perpetuity. Quantum BioPharma retains 100% of the rights to develop similar product or alternative formulations specifically for pharmaceutical and medical uses.
Forward-Looking Information
Certain information in this news release constitutes forward-looking statements under applicable securities laws. Any statements that are contained in this news release that are not statements of historical fact may be deemed to be forward-looking statements. Forward-looking statements are often identified by terms such as “planned”, “may”, “should”, “anticipate”, “expect”, “potential”, “believe”, “intend”, “targeting” or the negative of these terms and similar expressions.
Readers are cautioned that the foregoing list is not exhaustive. Readers are further cautioned not to place undue reliance on forward-looking statements, as there can be no assurance that the plans, intentions or expectations upon which they are placed will occur. Such information, although considered reasonable by management at the time of preparation, may prove to be incorrect and actual results may differ materially from those anticipated.
Forward-looking statements contained in this press release are expressly qualified by this cautionary statement and reflect the Company’s expectations as of the date hereof and are subject to change thereafter. The Company undertakes no obligation to update or revise any forward-looking statements, whether as a result of new information, estimates or opinions, future events or results or otherwise or to explain any material difference between subsequent actual events and such forward- looking information, except as required by applicable law.
Contacts:
Quantum BioPharma Ltd.
Zeeshan Saeed, Founder, CEO and Executive Co-Chairman of the Board
Email: Zsaeed@quantumbiopharma.com
Telephone: (416) 854-8884
Investor Relations
Email: ir@quantumbiopharma.com, info@quantumbiopharma.com
Website: www.quantumbiopharma.com
A photo accompanying this announcement is available at https://www.globenewswire.com/NewsRoom/AttachmentNg/e16adc7c-2200-4f53-bb33-b8dd37e4c1aa

Legal Disclaimer:
EIN Presswire provides this news content "as is" without warranty of any kind. We do not accept any responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you have any complaints or copyright issues related to this article, kindly contact the author above.